الوظائف ذات الصلة
جهاز الموجات فوق الصوتية لاستخراج البروتين: ميزة تحضير العينة في بيانات البروتين القابلة لإعادة الإنتاج
2026-03-25Protein Extraction Ultrasonicator workflows are gaining more attention because proteomics and molecular biology teams now treat sample prep as a key factor in data quality, not just a routine first step. For overseas buyers, distributors, and lab managers, that matters because poor upstream disruption can reduce reproducibility, increase rework, and weaken confidence in downstream results.
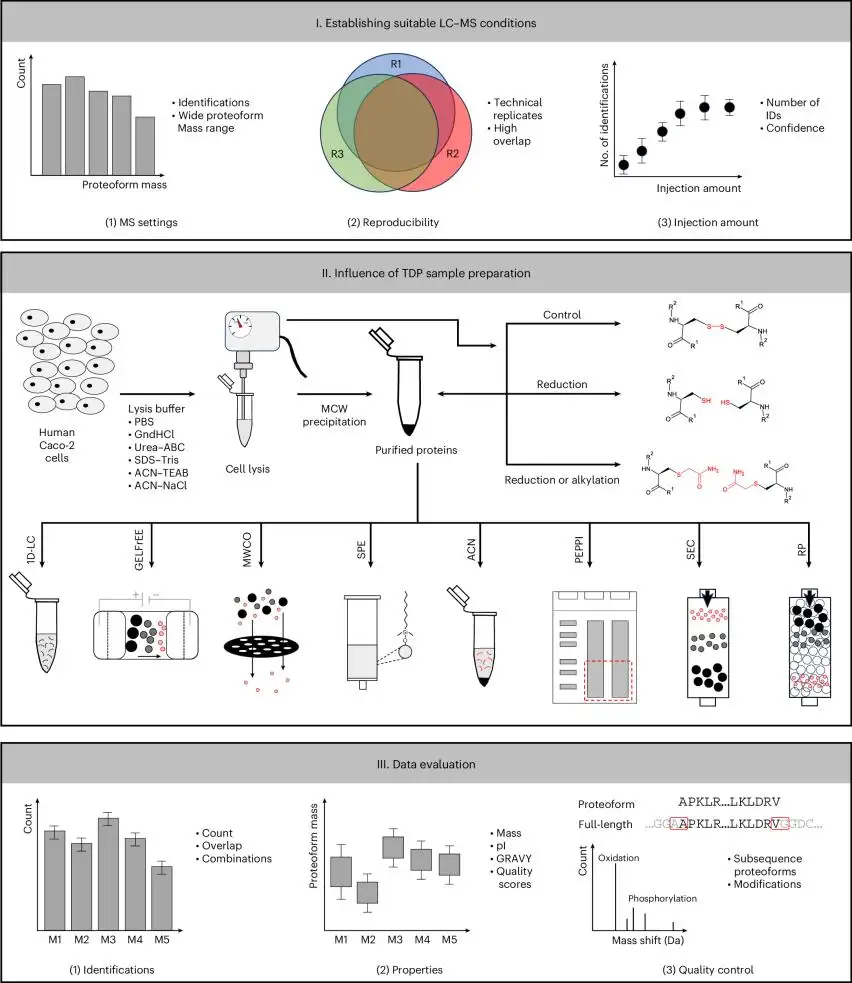
(Influence of different sample preparation approaches on proteoform identification by top-down proteomics)
Recent international research supports this shift. In 2025, L. S. Guadalupi and colleagues reported an ultrasound-assisted protein extraction workflow for microalgal samples designed to improve scalability and quantitative recovery from difficult biological matrices. Another 2025 study by D. Katsavelis and colleagues compared six serum proteomics sample-preparation workflows and showed that preparation strategy can directly influence quantitative performance and proteome coverage.
The Real Industry Problem Is Not Sonication Alone
Many laboratories still think the decision is simply about whether to sonicate or not. In practice, the bigger issue is how controlled the energy delivery is and how much operator variability the workflow still allows.
Traditional direct-contact sonication can create several familiar problems:
• Inconsistent energy transfer from sample to sample
• Higher contamination risk from probe contact
• Heat buildup that can compromise sensitive biomolecules
• More dependence on operator timing and handling
• Extra equipment footprint and workflow complexity
These are not small inconveniences. For procurement teams, they affect assay credibility, training burden, failure rates, and instrument utilization over time.
Recent protocol literature also points in the same direction. A 2025 dxChIP-seq protocol by Y. Bao and colleagues explicitly incorporated focused ultrasonication into a workflow designed to improve mapping of chromatin factors and signal-to-noise performance. That is important because it shows focused acoustic processing is not just a convenience feature. It is being built into newer high-value experimental workflows where fragmentation quality and reproducibility matter.
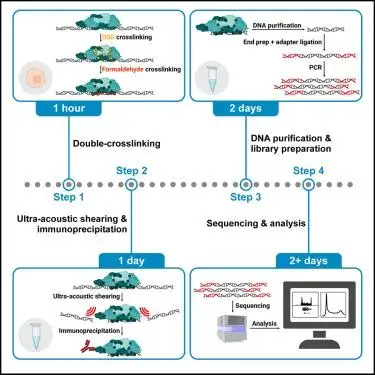
(Protocol for double-crosslinking ChIP-seq to improve data quality and enhance detection of challenging chromatin targets)
Why Focused Ultrasonication Fits Modern Protein Extraction Better
Longlight Technology's approach is based on focused, non-contact ultrasound delivery. In practical purchasing terms, that changes the workflow in several useful ways.
• Better Control Over Sample Processing
By concentrating high-frequency acoustic energy on the sample zone, the system helps reduce the broad energy dispersion that often makes bulk sonication harder to standardize. That supports more repeatable disruption across similar sample sets, especially where protein extraction quality must remain stable across operators or batches.
• Lower Contamination Probability
Because the acoustic energy is transmitted through a medium rather than through a probe that directly touches the sample, the workflow reduces one of the classic concerns in precious-sample handling: cross-sample contamination from direct-contact hardware. This matters in protein extraction, chromatin work, nucleic-acid workflows, and other high-sensitivity applications where trace contamination can distort results.

• True Low-Temperature Handling
Heat is one of the quietest causes of downstream inconsistency. Longlight's temperature sensing and control architecture, paired with integrated semiconductor cooling, is designed to keep processing conditions stable during ultrasonication. For labs working with temperature-sensitive proteins or mixed biomolecule workflows, that is a meaningful advantage over methods where thermal buildup is harder to manage.
• Easier SOP Standardization
The simpler the setup, the easier it is to train operators and maintain workflow consistency. Longlight's integrated design avoids reliance on an external computer and separate cooling hardware, which helps reduce bench clutter and setup variability. For laboratories building GMP-minded routines, shared-service workflows, or cross-team protocols, that can shorten SOP rollout and improve execution discipline.
What Recent Research Is Telling Buyers
The commercial takeaway from current research is straightforward: sample prep is now a performance decision, not just a preprocessing step.
The 2025 work by Guadalupi's team is a useful example because it targeted a difficult protein-extraction matrix and emphasized scalability and quantitative recovery. The 2025 serum proteomics comparison by Katsavelis and colleagues reinforces the same broader message from another angle: workflow design choices upstream materially shape quantitative outcomes downstream.
Even beyond proteomics, reproducibility is becoming a central purchasing criterion across sample-driven platforms. A 2025 Nature Biotechnology study led by J. T. Plummer and colleagues proposed standardized metrics and SOP-oriented assessment frameworks for reproducibility in spatial workflows, reflecting a wider market push toward measurable quality control and comparable technical performance.
(Standardized metrics for assessment and reproducibility of imaging-based spatial transcriptomics datasets)
For buyers, that means the evaluation standard should move beyond headline power or simple throughput claims. More useful questions include:
• Can the system reduce operator-dependent variation?
• Can it protect heat-sensitive samples during processing?
• Can it support both proteomics and adjacent workflows such as chromatin shearing or nucleic-acid extraction?
• Can it simplify training and documentation in a busy lab environment?
• Where Longlight Technology Creates Practical Value
Longlight Technology does not only address protein extraction. Its broader genomics and molecular biology positioning also matters for labs that want platform flexibility.
The same focused ultrasonication logic can support:
• اضطراب الخلايا والأنسجة لاستخراج الجينوم أو البروتين
• Fragmentation and homogenization of biological tissues
• DNA, RNA, and chromatin shearing
• FFPE-related preparation steps
• NGS-oriented fragmentation workflows
That wider applicability is commercially useful because many laboratories no longer buy instruments for a single isolated task. They look for workflow fit across proteomics, sequencing, molecular diagnostics, and translational research.
A Better Buying Standard for 2026
The strongest reason to consider a Protein Extraction Ultrasonicator today is not that ultrasound is new. It is that the market now expects cleaner sample handling, stronger repeatability, lower contamination risk, and more SOP-ready operation than older manual or direct-contact workflows typically deliver.
For distributors, procurement managers, and research teams, Longlight Technology's focused ultrasonication approach aligns well with that shift. Compared with traditional sonication methods, it offers a more controlled non-contact workflow, tighter temperature management, easier standardization, and better compatibility with modern multi-application labs. Supported by the direction of recent international research, that makes it a practical choice for laboratories that want more reliable upstream preparation before valuable downstream analysis begins.