الوظائف ذات الصلة
كريو-إم عالي الدقة: كيف تصل مشاريع علم الأحياء الهيكلي إلى خرائط عالية الجودة بشكل أسرع
2026-03-20High-Resolution Cryo-Em has become one of the most important tools in modern structural biology, but many projects still lose time and budget long before they reach a publishable 3D map. For overseas buyers, biotech teams, and research service users, the real bottleneck is often not access to a powerful microscope. It is whether the workflow can reliably screen sample quality, control risk early, and return complete data that scientists can trust for downstream interpretation.
(Scientists break resolution records to visualize individual atoms with single-particle cryo-EM)
That matters even more now because cryo-EM is no longer a niche method. The Electron Microscopy Data Bank had reached 55,737 entries as of March 25, 2026, showing how rapidly cryo-EM has expanded across structural biology, virology, and drug discovery. At the same time, recent market analyses continue to link cryo-EM demand to growing activity in structural biology, vaccine research, and structure-based drug development.
The Industry Pain Point Is Not Just Resolution
A common assumption in procurement is that better hardware automatically leads to better structure results. In practice, that is incomplete.
Many cryo-EM projects slow down because of issues that appear much earlier in the pipeline:
• Sample heterogeneity
• Aggregation or poor particle distribution
• Weak screening logic before expensive data collection
• Incomplete processing support
• Limited transparency in data delivery and validation
This is exactly why cryo-EM has gained ground over X-ray crystallography in so many research programs. It needs less sample, does not require crystallization, preserves biomolecules closer to their native state, and can handle difficult targets such as membrane proteins, flexible assemblies, and viral particles. Those advantages are well established, but they only translate into real project success when the service workflow is disciplined from the first screening step.
Recent Research Shows Why Better Front-End Workflow Matters
The latest international studies make this point very clearly.
In 2025, Willy W. Sun, Dennis J. Michalak, Justin W. Taraska and colleagues reported a cryo-electron tomography pipeline for plasma membranes in Nature Communications. The work addressed one of the field's hardest problems: imaging membrane proteins in native cellular contexts. A related Nature Reviews Bioengineering analysis noted that plasma membrane cryo-ET remains challenging because samples must be thinner than 300 nm, while common thinning methods can be low throughput and still produce limited resolution. In other words, advanced cryo-EM studies depend heavily on sample preparation strategy and screening quality, not just on microscope access.
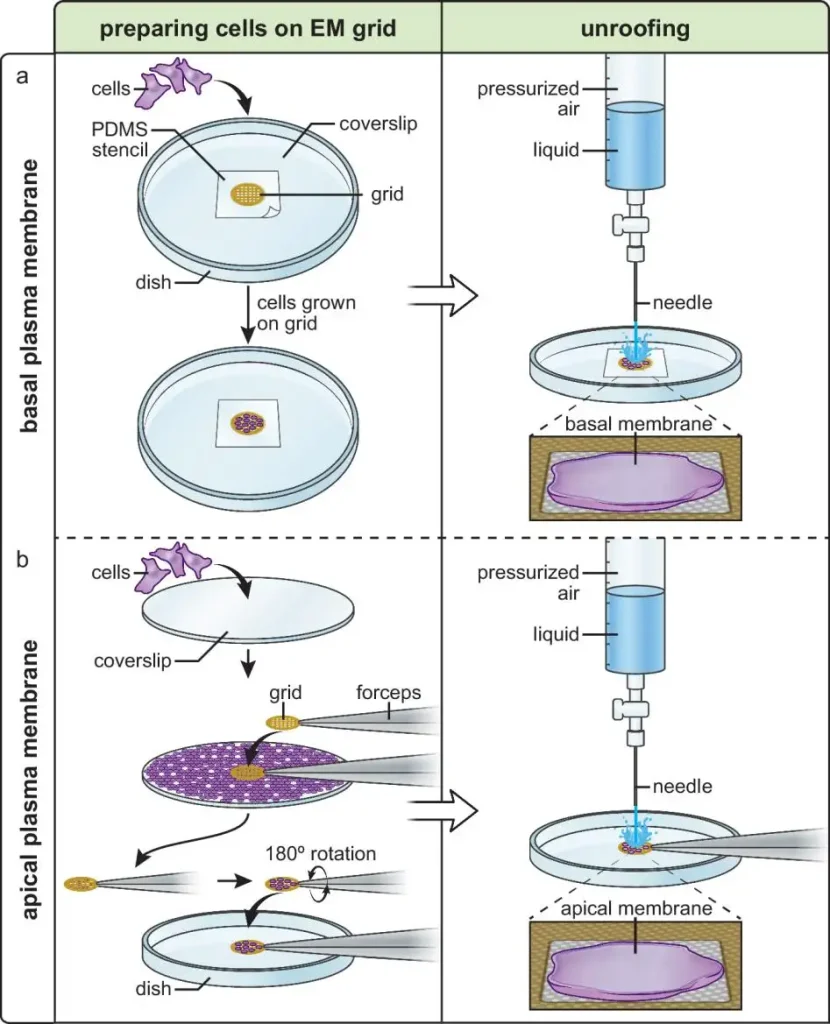
(Cryo-electron tomography pipeline for plasma membranes | Nature Communications)
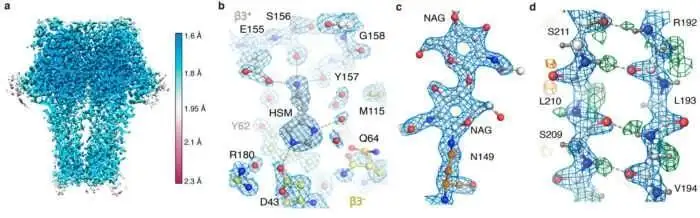
Another strong example came from Daniel B. Haack and colleagues in Nature Communications. Their 2025 study on scaffold-enabled high-resolution cryo-EM structure determination of RNA showed that small and previously difficult RNA targets could be resolved at 2.5 Å when the workflow was engineered correctly. The paper is important for buyers because it highlights a core commercial lesson: many "hard targets" are not impossible targets. They require better experimental design, better screening logic, and better downstream reconstruction support.
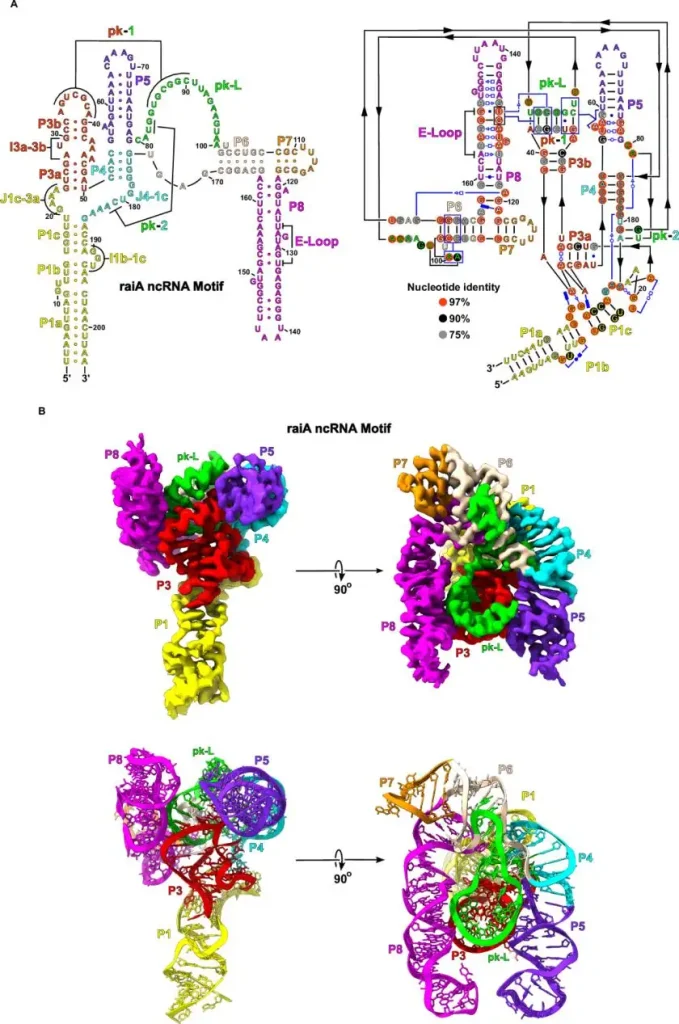
(Scaffold-enabled high-resolution cryo-EM structure determination of RNA | Nature Communications)
These studies reflect what many laboratories already feel in practice: the challenge in High-Resolution Cryo-Em is no longer only imaging power. It is workflow reliability from sample receipt to map validation.
Where Longlight Technology Fits Into This Need
For research groups and commercial users, Longlight Technology addresses this gap by organizing cryo-EM services around risk control, staged decision-making, and full-result transparency.
Instead of pushing directly into expensive high-resolution collection, Longlight starts with sample suitability assessment through negative stain screening. This helps evaluate homogeneity, aggregation, morphology, size, and distribution before more resources are committed. For buyers, that approach is valuable because it reduces avoidable instrument time and lowers the chance of moving weak samples into a costly acquisition stage.
From there, Longlight supports two major structural routes:
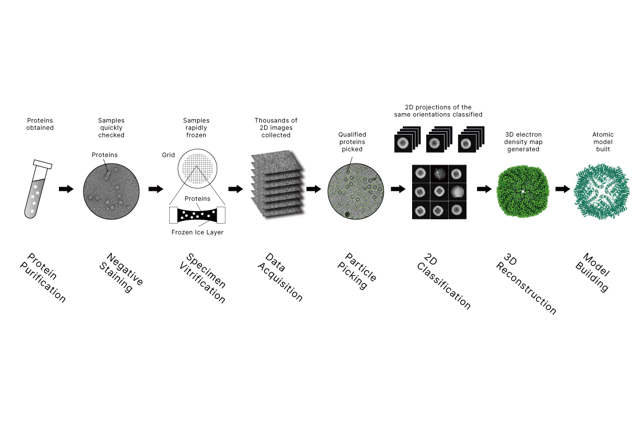
• Single Particle Analysis For High-Resolution Reconstruction
For soluble proteins, membrane proteins, and supramolecular assemblies, Longlight supports a single-particle workflow that converts large sets of 2D particle projections into 3D density maps and, where applicable, refined atomic models. This is especially relevant for teams working on biologics, antibody programs, viral targets, and structure-guided discovery.
• Cryo-Electron Tomography For Native Context
When the research question is spatial rather than purely isolated-structure based, cryo-ET provides a better path. It is suited to membrane-associated complexes, viral infection studies, and large biological assemblies in native contexts. That makes it a strong option for projects similar to the 2025 plasma membrane work discussed above.
لماذا It Is Stronger Than Traditional Structural Routes
Compared with more traditional structure workflows, a mature cryo-EM service model offers practical advantages that overseas buyers care about:
• No crystallization requirement, which reduces delays for difficult targets
• Near-native preservation, which supports biologically relevant interpretation
• Conformation heterogeneity analysis, useful for dynamic complexes
• Lower sample burden, especially valuable for precious materials
• Broader target compatibility, including membrane proteins and viral assemblies
Longlight strengthens these natural cryo-EM advantages by adding service-level benefits that are often overlooked in vendor comparisons: full data delivery, intermediate processing files, final density maps, atomic coordinate models when applicable, and validation outputs such as MolProbity-related assessments. That level of transparency is important for research teams that need internal review, publication support, IP documentation, or multi-site collaboration.
A More Practical Buying Standard for Cryo-EM Services
For procurement managers and technical decision-makers, the better question is not simply, "Who has access to advanced cryo-EM instruments?" The better question is, "Who can reduce failure risk across the entire project?"
A reliable High-Resolution Cryo-Em partner should be able to offer:
• Sample quality inspection before major instrument commitment
• Clear workflow from consultation to final report
• Support for both screening and high-resolution reconstruction
• Cryo-ET capability for in situ biology questions
• Complete and portable data delivery for long-term reuse
This is where Longlight Technology stands out. By combining structured screening, advanced cryo-EM workflows, experienced processing support, and full-result transparency, it helps research teams move from uncertain samples to decision-ready structural data with greater efficiency.
In today's structural biology market, that is the real competitive advantage. High resolution still matters, but the teams that reach it faster are usually the ones that manage the workflow better from day one.