الوظائف ذات الصلة
الموجات فوق الصوتية المركزة لأنسجة النبات: لا تزال الوحدات متعددة الأوميكس في النبات تبدأ بمشكلة تحضير العينة
2026-03-19Focused Ultrasonicator for Plant Tissues is becoming more relevant in plant genomics and epigenomics because many labs no longer struggle with sequencing access first—they struggle with whether tough, variable plant samples can be processed into clean, reproducible input for sequencing, ChIP-seq, and extraction workflows.
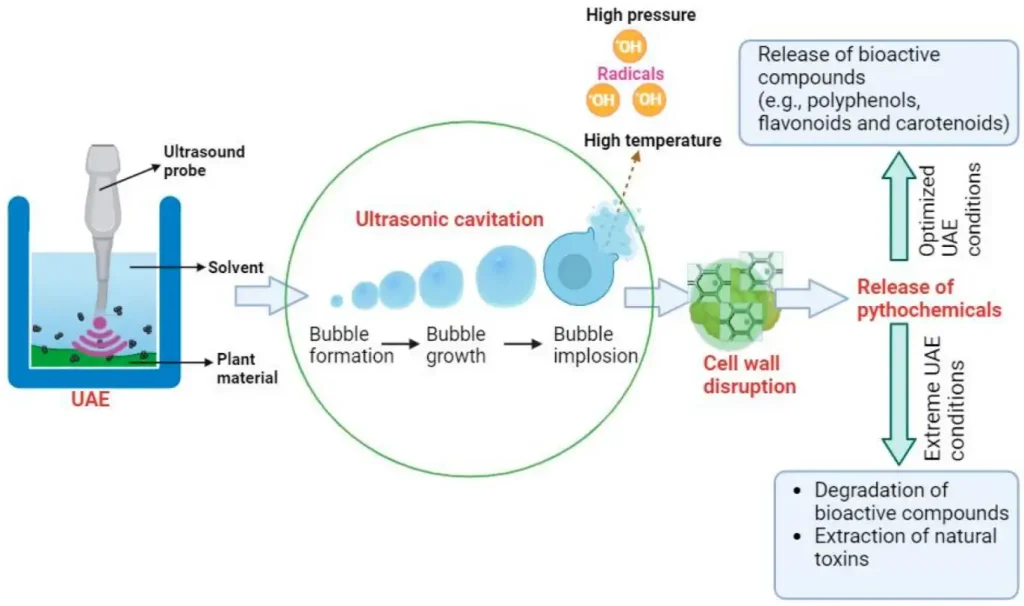
(Overview and Toxicity Assessment of Ultrasound-Assisted Extraction of Natural Ingredients from Plants)
That bottleneck matters more than many buyers expect. Leaves, stems, reproductive tissues, and stress-treated plant materials are not easy samples. Cell walls are rigid. Secondary metabolites interfere with extraction. Heat buildup during disruption can damage nucleic acids or distort downstream consistency. In busy research labs or commercial breeding programs, these issues show up as poor repeatability, inconsistent fragment size, failed libraries, and more time spent re-running samples than analyzing data.
Why Plant Tissue Preparation Remains a Pain Point
Plant workflows are uniquely demanding because sample heterogeneity is built into the biology. A young Arabidopsis seedling, a lignified stem section, and a reproductive tissue sample do not behave the same way during disruption or chromatin processing. That makes manual, contact-based, or heat-prone approaches especially risky.
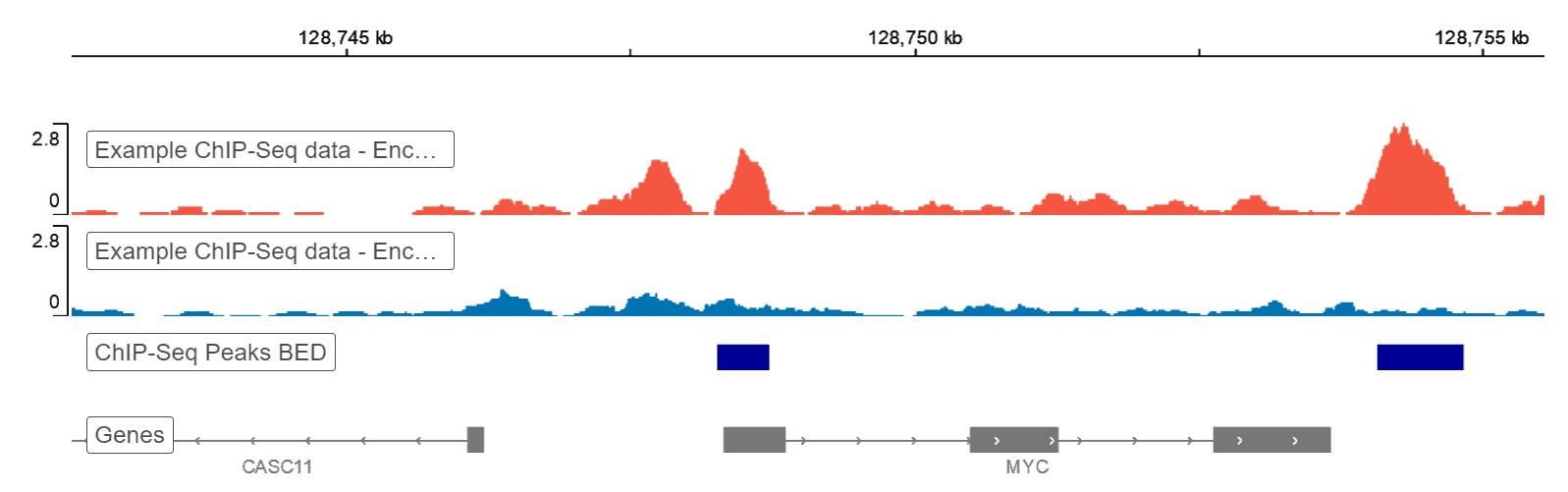
A 2024 study introducing PHILO (Plant HIgh-throughput LOw input) ChIP-seq, led by A. Choudhary and colleagues, described plant chromatin profiling itself as a scalability challenge and noted that established plant ChIP-seq methods are difficult to expand because of the substantial sample volumes and fragmentation burden involved. Their platform was designed to process more than 100 samples in parallel, with gains in scalability, lower input requirements, user-friendliness, and cost efficiency over conventional workflows.
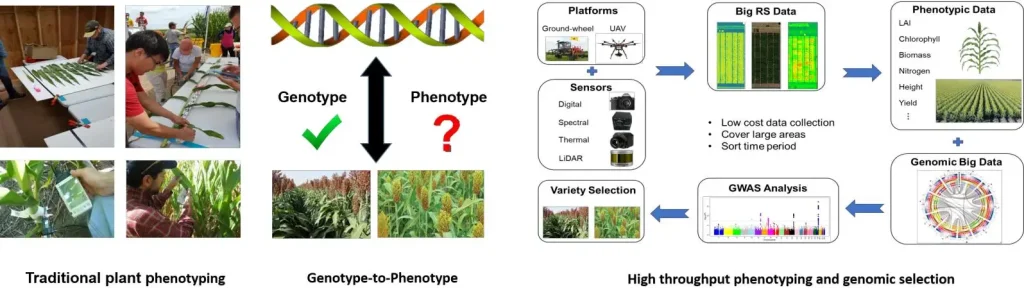
(High throughput plant phenotyping)
That is exactly why sample disruption and fragmentation hardware matters. When high-throughput plant workflows grow, the weak point often shifts upstream:
• Inconsistent tissue disruption
• Operator-to-operator variability
• Contamination risk in direct-contact systems
• Thermal damage during prolonged processing
• Fragmented lab layouts that require external cooling and PC control
These are not minor convenience issues. They influence data credibility, throughput planning, and total operating cost.
What Recent Research Says About Better Plant Sample Processing
One useful reference comes from a plant DNA extraction study published in Applications in Plant Sciences by Alexia Stettinius and co-authors. The team evaluated focused ultrasound extraction (FUSE) on leaf tissues from American chestnut, tulip poplar, red maple, and chestnut oak. They reported DNA extraction in 9–15 minutes, versus 30 minutes for control extraction methods, and showed that the released DNA was suitable for amplification and next-generation sequencing. In two species, DNA yield was also substantially higher than the control workflow.
Another strong signal comes from the plant epigenetics side. In a 2025 Nature Cell Biology paper, Guanghui Xu, Julie A. Law, and colleagues showed that tissue-specific DNA methylation in Arabidopsis reproductive tissues is directed by transcription factors and sequence features, helping explain how distinct epigenomes are generated in anthers and ovules. That kind of work depends on reliable handling of delicate plant tissues and chromatin-linked material, because tissue-specific epigenetic signals are easy to blur when preparation steps introduce heat, over-processing, or poor reproducibility.
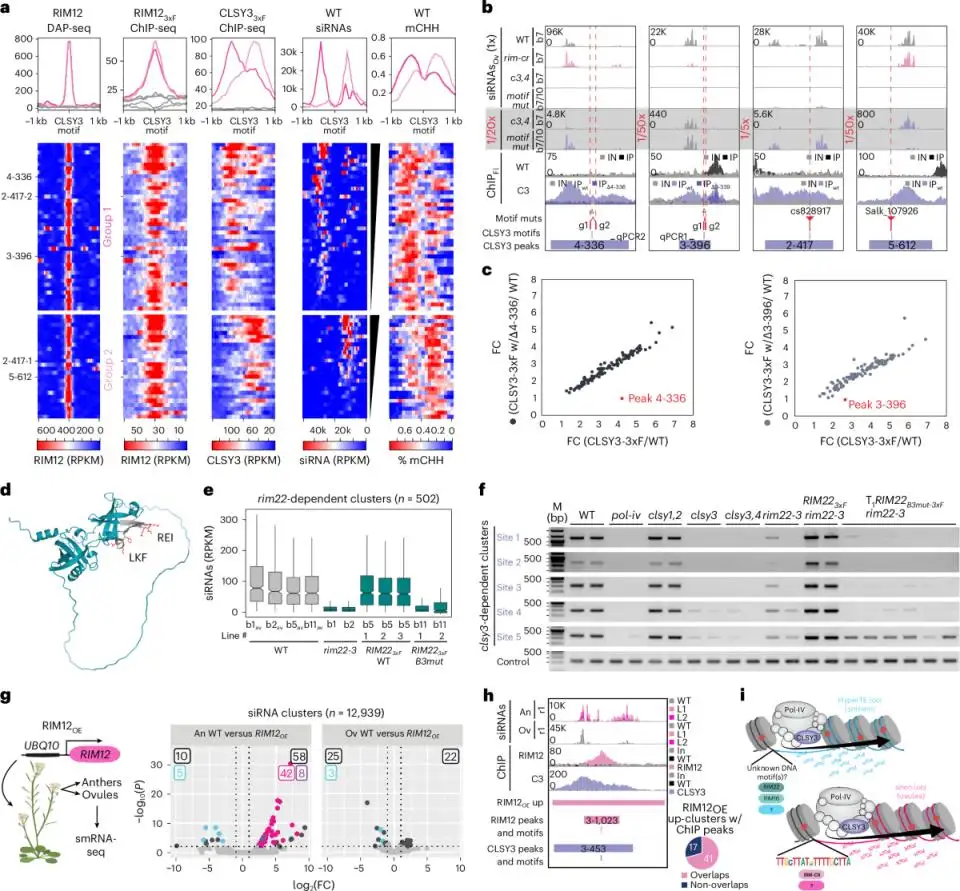
(Transcription factors instruct DNA methylation patterns in plant reproductive tissues)
Together, these studies point to the same purchasing lesson: plant research is moving toward higher sensitivity, lower input, and higher throughput, so sample preparation systems must become more controlled, not more improvised.
How Longlight Technology Fits This Shift
This is where Longlight Technology has a clear positioning advantage. Its focused ultrasound platform is designed around controllability rather than brute-force disruption. For plant tissue workflows, that matters.
Based on the specifications you shared, the system offers several practical strengths:
• Non-contact sample processing, which lowers contamination risk compared with direct-contact sonic crushing systems
• True low-temperature and constant-temperature control, supported by high-sensitivity sensing and a built-in semiconductor refrigeration system
• Focused acoustic energy, which improves process control and repeatability
• Integrated design, with no need for an external computer or separate cooling module
• Simple parameter-based operation, which reduces training burden and manual inconsistency
• Quiet operation, making it easier to deploy in shared lab environments
For buyers building genomics or molecular biology workflows, this means Longlight is not only selling an instrument. It is reducing common pre-analytical friction.

Why Focused Ultrasound Has an Edge Over Traditional Methods
Traditional disruption methods often do the job, but not always with the level of control that current plant genomics projects demand. Contact-based systems can raise contamination concerns. Traditional bulk sonication may lack precise energy uniformity, and hands-on handling can make experimental results more sensitive to operator practice. External chillers and computer-linked setups also add bench complexity.
A focused system changes that value proposition in several ways.
First, sample integrity improves because the workflow is more temperature-stable. That is especially useful for DNA, RNA, and chromatin-related applications where overheating can erode downstream performance.
Second, repeatability improves because acoustic delivery is more standardized. For labs running ChIP-seq, NGS fragmentation, genome extraction, or tissue homogenization, that can translate into more predictable downstream behavior.
Third, workflow efficiency improves because integrated cooling and standalone operation reduce setup dependencies. In practical purchasing terms, that means less equipment sprawl and less coordination between separate modules.
For plant labs comparing options, the advantage is not simply "stronger disruption." It is cleaner, more standardized disruption.
Why This Matters for Buyers أناn 2026
The commercial backdrop also supports this shift. Mordor Intelligence estimates the plant genomics market at USD 8.5 billion in 2025, growing to USD 15.2 billion by 2030 at a 12.3% CAGR, with DNA sequencing still the cornerstone technology. The same report ties growth to falling sequencing costs and wider adoption across breeding pipelines.
Its agrigenomics analysis also notes that the broader market is expected to reach USD 5.49 billion in 2026, while NGS continues to gain momentum because of large data output, multi-omics integration, and AI-driven analytics.
That means procurement decisions are changing. Buyers are no longer selecting instruments only for isolated experiments. They are selecting platforms that can support:
• Breeding genomics
• Plant epigenetics
• ChIP-seq and chromatin studies
• NGS library preparation
• Tissue disruption for DNA, RNA, and protein extraction
• More standardized multi-user laboratory workflows
In that environment, a Focused Ultrasonicator for Plant Tissues is valuable when it helps laboratories reduce variability before the sequencing step even begins.
The Practical Takeaway
For plant research, the sample prep problem is no longer hidden. It is now one of the most visible reasons why otherwise well-funded workflows still lose time, consistency, and confidence.
Longlight Technology addresses that problem with a focused, non-contact, temperature-controlled ultrasound approach that aligns well with where plant genomics is heading: lower input, higher reproducibility, cleaner workflows, and better support for advanced plant DNA, RNA, chromatin, and tissue-processing applications.
For international buyers, that is the stronger purchasing argument. The right platform does not just process plant tissue. It helps make downstream data more trustworthy.