الوظائف ذات الصلة
جهاز القص بالموجات فوق الصوتية للحمض النووي: كيف يحسن MALDI-TOF الكشف عن الميكروبات التي يصعب تحليلها
2026-03-23DNA Shearing Ultrasonicator is becoming highly relevant far beyond genomics, because the same core issue also limits microbial protein extraction: when sample disruption is inconsistent, even a powerful MALDI-TOF workflow can lose speed, confidence, and identification depth before the spectrum is ever generated. MALDI-TOF MS has already transformed clinical microbiology by cutting identification time versus conventional biochemical workflows, but difficult organisms such as mycobacteria, Nocardia, and filamentous fungi still expose a weak point in many labs: pretreatment is often too manual, too variable, and too dependent on large biomass input.
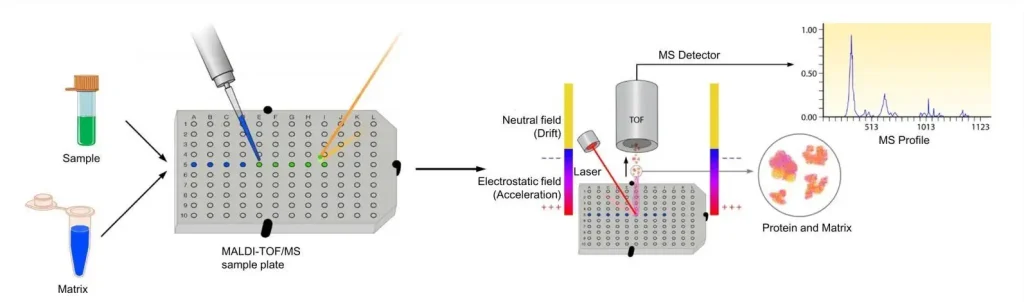
(MALDI-TOF Mass Spectrometry - Creative Proteomics)
The Real Bottleneck Is Not the MALDI-TOF Instrument
For many overseas buyers, the purchasing question is no longer whether MALDI-TOF is fast. It is whether the upstream workflow can prepare challenging samples fast enough, safely enough, and reproducibly enough to let the platform deliver its true value. Recent reviews of clinical microbiology mass spectrometry make this point clear: MALDI-TOF is now a mature identification technology, yet sample preparation remains one of the most decisive factors affecting routine performance, especially in complex or difficult-to-disrupt organisms.
That issue matters even more in mycobacterial testing. The WHO's latest global tuberculosis reporting shows that TB remains a major international diagnostic burden, which means faster and more dependable organism handling still matters at scale. At the same time, mycobacterial and mold identification continue to be listed among the more challenging use cases for MALDI-TOF in routine laboratories.
Why Hard-To-Lyse Organisms Still Reduce Detection Confidence
In practice, difficult microorganisms create a familiar chain of problems:
• Too much colony material is needed to get enough extractable protein
• Ethanol, formic acid, acetonitrile, or bead-beating workflows add steps and handling time
• Repeated tube opening increases contamination and operator exposure risk
• Manual pretreatment creates run-to-run variability
• Weak or incomplete protein release can reduce identification confidence
These are not minor inconveniences. They affect throughput, biosafety, and the economic return of the MALDI-TOF platform itself. A 2025 paper by A. M. Bazzi and colleagues showed that the direct identification of Mycobacterium species using MGIT liquid medium delivered 88.6% overall accuracy. The study nevertheless suggested that more optimization is needed to improve results for rarer species. That is a useful signal for procurement teams: the field is advancing, but better front-end disruption and extraction remain important.
(Rapid identification and drug susceptibility screening of ESAT-6 secreting Mycobacteria by a NanoELIwell assay)
A second 2025 study by Ö. Dogan and colleagues focused on filamentous fungi and showed that workflow choice still has a major impact on outcome. Their comparative analysis reported that a time-efficient workflow combining optimized extraction conditions and database selection reached 94.1% accuracy for filamentous fungi, underlining how strongly identification performance depends on preparation strategy rather than instrument branding alone.
How Longlight Technology Helps Solve This Workflow Gap
This is where Longlight Technology becomes strategically relevant. Its focused ultrasound approach is designed to improve sample pretreatment by concentrating acoustic energy precisely into the sample zone rather than dispersing it broadly like traditional water-bath sonication or relying on direct-contact probe systems. In practical terms, that supports faster cell disruption, better extractant penetration, and more efficient protein release from hard-to-break organisms. The result is not just speed. It is a more controlled path to usable spectra.
For labs working with difficult bacterial colonies, mycobacteria, Nocardia, spores, or filamentous fungi, Longlight's technology offers several workflow advantages:
• Closed-tube processing that reduces repeated lid opening
• Faster pretreatment, with ultrasound completed in about 60 seconds and full workflow finished in under 5 minutes according to the product description
• Useful extraction from very small biomass input, including only 1–2 colonies in some hard-to-lyse organisms
• Non-contact, isothermal water-bath processing that helps reduce heat-related variability
• More standardized output through calibrated acoustic delivery and temperature control
These advantages align closely with what current literature says labs still need: less manual variation, less operator-dependent pretreatment, and better consistency across routine runs.
Why Focused Ultrasound Is More Practical Than Traditional Disruption Methods
Traditional pretreatment methods still work, but they often force labs to trade one problem for another. Probe ultrasound can introduce contamination concerns and uneven exposure if workflows are not tightly controlled. Water-bath methods can lose energy efficiency. Multi-step chemical extraction can be effective, but it increases handling burden and extends turnaround time. For procurement managers and laboratory directors, this becomes a hidden cost center: more labor, more retraining, more repeat work, and more uncertainty on difficult samples.
تقنية Longlight's focused ultrasound design changes that equation by making energy delivery more localized and reproducible. Instead of relying on less controlled disruption formats, it gives laboratories a more standardized and scalable way to prepare difficult samples.

Key advantages include:
• More localized acoustic energy delivery, which helps improve processing consistency
• Closed-tube, non-contact operation, which makes workflows easier to standardize across different operators
• Temperature-controlled processing, which is better suited to sensitive nucleic acid and protein preparation tasks
• Reduced manual variability, helping labs improve repeatability in routine sample pretreatment
• Broader application value, allowing one platform to support multiple laboratory workflows
The same technology can be used for:
• Cell lysis
• Proteomics
• Chromatin workflows
• DNA/RNA shearing
• FFPE extraction
• تحلل الكائنات الدقيقة الصعبة
For buyers, that means one pretreatment investment can support both diagnostic and research workflows. This kind of versatility is especially valuable for laboratories that want to improve workflow efficiency without adding multiple specialized systems.
What Overseas Buyers Should Really Evaluate
When international buyers compare pretreatment systems for MALDI-TOF-related workflows, the key question is not simply "How strong is the instrument?" A better question is: "How reliably does it turn a hard sample into a high-confidence input for downstream analysis?"
A strong evaluation framework should include:
• Performance on hard-to-lyse organisms
• Time from colony to ready-to-test extract
• Closed-tube biosafety and contamination control
• Sample input requirements
• Reproducibility across operators and shifts
• Flexibility for protein and nucleic acid workflows beyond microbiology
That is why the conversation around DNA Shearing Ultrasonicator should not be limited to DNA fragmentation alone. In the hands of a supplier like Longlight Technology, focused ultrasonics becomes a broader sample-preparation solution that supports faster, safer, and more standardized pretreatment for the kinds of difficult organisms that still challenge MALDI-TOF laboratories worldwide. The instrument at the end of the workflow may generate the spectrum, but pretreatment still decides how much useful biology reaches it.